
For thermal ablation of saphenous veins
This wavelength of 1470 nm combined with its “continuous” firing mode are now recognized as the Gold Standard in the thermal ablation treatment of saphenous by laser.
All national and international recommendations are at the origin of the recognition of this Gold Standard:
Internationally:
- ESVS 2022: See more
- USA:
- Revue Cochrane:
Why the wavelength 1470 nm?
a) Selective energy delivery
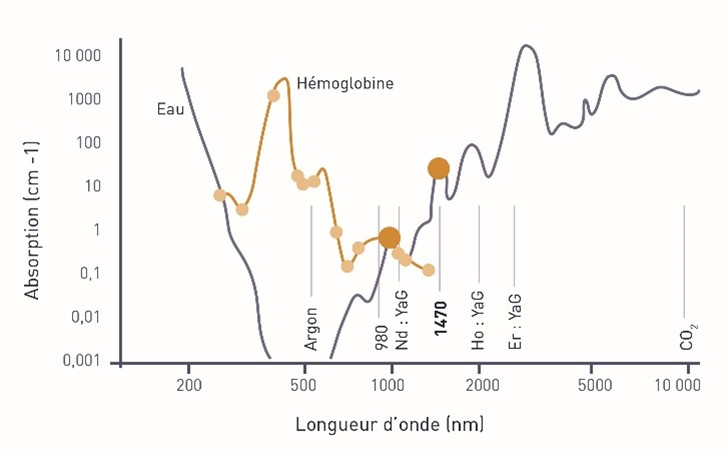
The Endotherme™ 1470 laser emits infrared light with a wavelength of 1470 nm.
Emitted inside the saphenous, the light is thus absorbed by the water (its chromophore) of the venous wall and then converted into heat.
Under the effect of this heat, the venous wall is destroyed, which leads to the occlusion of the saphenous.
b) Focused energy delivery
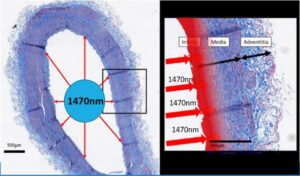
The penetration distance of the wavelength 1470 is of the order of 300 to 400 μm. During thermal endovenous treatment, the 1470 laser acts on more than half the thickness of the venous wall (thickness between 450 and 600 μm), inducing a level of tissue damage necessary (to induce the destruction of the vein) and sufficient (not to induce damage in the adjacent compartment).
c) Conclusion
Wavelength Diode Laser 1470 nm + Radial Emission Fibers:
- Low light penetration (0.34mm) by WL 1470 nm
- Light oriented towards the venous wall by the radial emission fiber
- Short exposure time (thanks to continuous shrinkage)
=> Destruction of the Wall without destruction of surrounding tissues or nerves
Laser Endotherme™ 1470 – Indications
The precise targeting of the 1470 nm Endovenous Laser allows a high precision of the treatment as well as wide indications.
The Endovenous Laser is indicated for:
- Large saphenous veins (between 4 and 20 mm in diameter in orthostatic position)
- Small saphenous veins (between 4 and 20 mm in diameter in orthostatic position)
Laser Endotherme™ 1470 – Les contraindications
Endotherme™ 1470 should not be used in the following cases:
- Patients with thrombi in the venous segment intended for treatment.
- Patients with an aneurysm in the venous segment intended for treatment.
- Patients with peripheral vascular diseases with an ankle-arm systolic pressure index below 0.9.
- Patients with deep vein thrombosis or with a history of deep vein thrombosis.
- Pregnant patients.
- Patients with infection on the area to be treated, active herpes or other viral infection.
- Other contraindications may be indicated by the practitioner at the time of treatment.
Traitement endoveineux thermique – Le matériel
LSO Medical offers you a turnkey solution to launch a thermal endovenous activity.
You want to discuss your business project:
a) The 1470 nm Endotherme™ laser
Technical specifications:
- Laser: Laser Diode
- Wavelength: 1470 nm +/- 10
- Power: 15 Watts
- Cooling: Peltier and Air
- Continuous or Pulsed, 0.1 to 25 sec.
- Laser class: 4
- Class DM: IIb (Directive 93/42/EC)
- Aiming laser: Laser Diode
- Wavelength: 635 nm +/- 20
- Power: Less than 2 mWatts
- Laser class: 3R
- Power supply: 100-230 VAC; 50 to 60 Hz
- Weight: 15 kg
- Dimensions: H45 cm × L45 cm × W35 cm
b) Practical guide to use
Endovenous laser treatment Endotherme™ 1470:
- The 10-step procedure
- Material selection: Fiber and consumables
- How the Endotherme™ 1470 laser works
| Description | Reference | Technical specifications | Compatibility |
|---|---|---|---|
| Endotherme™ 1470 laser | PHARAON 1470 |
| Ringlight® fibers compatibility |
| Description | Reference | Technical specifications | Compatibility |
|---|---|---|---|
| RINGLIGHT® FIBER PROBE IR 1,8 mm | ORLF000003 | Ringlight® Fiber Standard 600 µm, 1.8 mm | Laser : Endotherme™ 1470 Introducer 6 Fr |
| RINGLIGHT® FIBER PROBE IRH 1,0 mm | ORLF000005 | Ringlight® Fiber Slim 400 µm, 1.0 mm | Laser : Endotherme™ 1470 Introducer 4 Fr or Cathlon 16G |
| RINGLIGHT® FIBER PROBE IR 1,8 mm | ORLF000007 | Ringlight® Fiber Fused Standard 600 µm, 1.8 mm | Laser : Endotherme™ 1470 Introducer 6 Fr |
| RINGLIGHT® FIBER PROBE IRH 1,3 mm | ORLF000006 | Ringlight® Fiber Fused Slim 400 µm, 1.3 mm | Laser : Endotherme™ 1470 Introducer 4 Fr or Cathlon 16G |
| Description | Reference | Technical specifications | Compatibility |
|---|---|---|---|
| Servante Endotherme™ 1470 | SERVANTE | Compatible Endotherme™ 1470 | |
| Lunettes Praticiens | FG1#37 | Blue glasses lenses | |
| Lunettes Patients | IRD5#35 | Green glasses lenses |
| Description | Reference | Technical specifications | Compatibility |
|---|---|---|---|
| DP 30 Pump – Nouvag | 4187 |
| Nouvag 6022 / 6022a / 6022b tubing |
| Tubing – Nouvag | 6022 / 6022a / 6022b | Length: 4 meters | DP 30 Pump – Nouvag |
| Vascular thermal ablation kit 6 Fr – LSO Medical | FR-K66189 | Ringlight® Fiber Standard | |
| Introduction kit 6 Fr – SCW | INTRO6F03818GS |
| Ringlight® Fiber Standard |
Destinations: vascular surgeons, vascular doctors, pharmacists, biomedical engineers and operating room staff.
Medical equipment Laser: Endotherme™ & LumeSeal_SB®. DM class: IIb. Laser class: 4
Disposables: Ringlight® and Ringlight_SB® Laser Fibers, DM Class: IIa.
![]() 1639
1639
Laser Disposables: Ringlight® and Ringlight_SB® fibers are sold sterile. They are sterilized with ethylene oxide. They are designed for single use only. They must not be reused or resterilized.
Endovenous Laser Devices:
Intended Use
The LumeSeal_SB® laser device is used to produce laser energy with controlled parameters (notably wavelength, power, and emission mode) and to deliver it to a compatible output device, such as an endovenous optical fiber, to allow the controlled application of thermal energy within the target vein during endovenous procedures. The device is used under ultrasound guidance and in accordance with the manufacturer’s instructions for use.
Intended Purpose
The LumeSeal_SB® laser device is intended to generate and deliver laser energy for therapeutic purposes, for endovenous use in the coagulation, occlusion, and ablation of the saphenous veins of the lower limbs in patients with chronic venous disease, when endovenous thermal treatment is clinically indicated. The device is intended for use in adult patients by trained healthcare professionals in an appropriate medical environment.
Indications for Use
The LumeSeal_SB® laser device is indicated for use in procedures for the coagulation, occlusion, and endovenous thermal ablation of saphenous veins in patients presenting with venous insufficiency associated with documented venous reflux. The indication is based on a prior clinical and hemodynamic assessment, including an echo-Doppler examination, and on the practitioner’s decision in accordance with current clinical recommendations.
Sterile Consumables:
Intended Use
The Ringlight® / Ringlight_SB® endovenous optical fiber is used to deliver, diffuse, and apply laser energy in a controlled manner within the target vein during endovenous procedures, according to the parameters defined by the compatible laser device. The fiber is used under ultrasound guidance and in accordance with the manufacturer’s instructions for use.
Intended Purpose
The endovenous optical fiber is intended to transmit and diffuse laser energy, generated by a compatible laser device, for therapeutic purposes, for endovenous use in the coagulation, occlusion, and ablation of the saphenous veins of the lower limbs in patients with chronic venous disease, when endovenous thermal treatment is clinically indicated. The endovenous optical fiber is intended for use in adult patients by trained healthcare professionals in an appropriate medical environment.
Indications for Use
The endovenous optical fiber is indicated for use in procedures for the coagulation, occlusion, and endovenous thermal ablation of the saphenous vein in patients presenting with venous insufficiency associated with documented venous reflux. The indication is based on a prior clinical and hemodynamic assessment, including an echo-Doppler examination, and on the practitioner’s decision in accordance with current clinical recommendations.
Benefits: The benefits of endovenous laser treatment are a size reduction or a complete obliteration of the treated varicose veins, and the improvement or even the resolution of venous symptoms. The risk of varicose veins complications will decrease significantly.
Contraindication: LumeSeal_SB® should not be used in the following cases: Patients with thrombi in the venous segment intended for treatment, Patients with an aneurysm in the venous segment intended for treatment, Patients with peripheral vascular diseases with an ankle-arm systolic pressure index below 0.9, Patients with deep vein thrombosis or with a history of deep vein thrombosis, Pregnant patients, Patients with infection on the area to be treated, active herpes or other viral infection, Other contraindications may possibly be indicated by the practitioner at the time of treatment. Read the instructions in the user manual carefully.
Adverse Effects: Based on the analyzed literature, three main categories of adverse effects can be identified for the laser procedure of GSV occlusion: Common adverse effects (incidence < 50%): bruising, pain, and superficial vein thrombosis. Rare adverse effects (incidence < 5% in most studies): deep vein thrombosis, paresthesia, and skin burns. Exceptional adverse effects (fewer than 10 cases reported): persistent paresthesia (lasting more than one year) and pulmonary embolism.
Device Use: Training in the use of LumeSeal® provided by LSO Medical or by individuals authorized by LSO Medical. Carefully read the instructions in the user manual. For the SnakeBack system to comply with the defined requirements, an annual replacement of the wheels and recalibration of the device must be performed, at least once a year. This maintenance must be carried out by a technician that has been officially certified by LSO MEDICAL or by its authorized local distributor. The non-compliance of this rule leads to the cancellation of the manufacturer guarantee.
Endovenous Laser Treatment: Covered by Health Insurance
Manufacturer: LSO Medical, Biocentre A. Fleming, Bât D – 280 rue Salvador Allende – 59120 Loos, France
Distributor and importer: SCW, NOUVAG
Internal reference: 23/04/LSOMEDICAL/PM/001
Version: IM EMB – VA-en


